Hidden Causes of Thyroid Symptoms Explained
Quick Answer
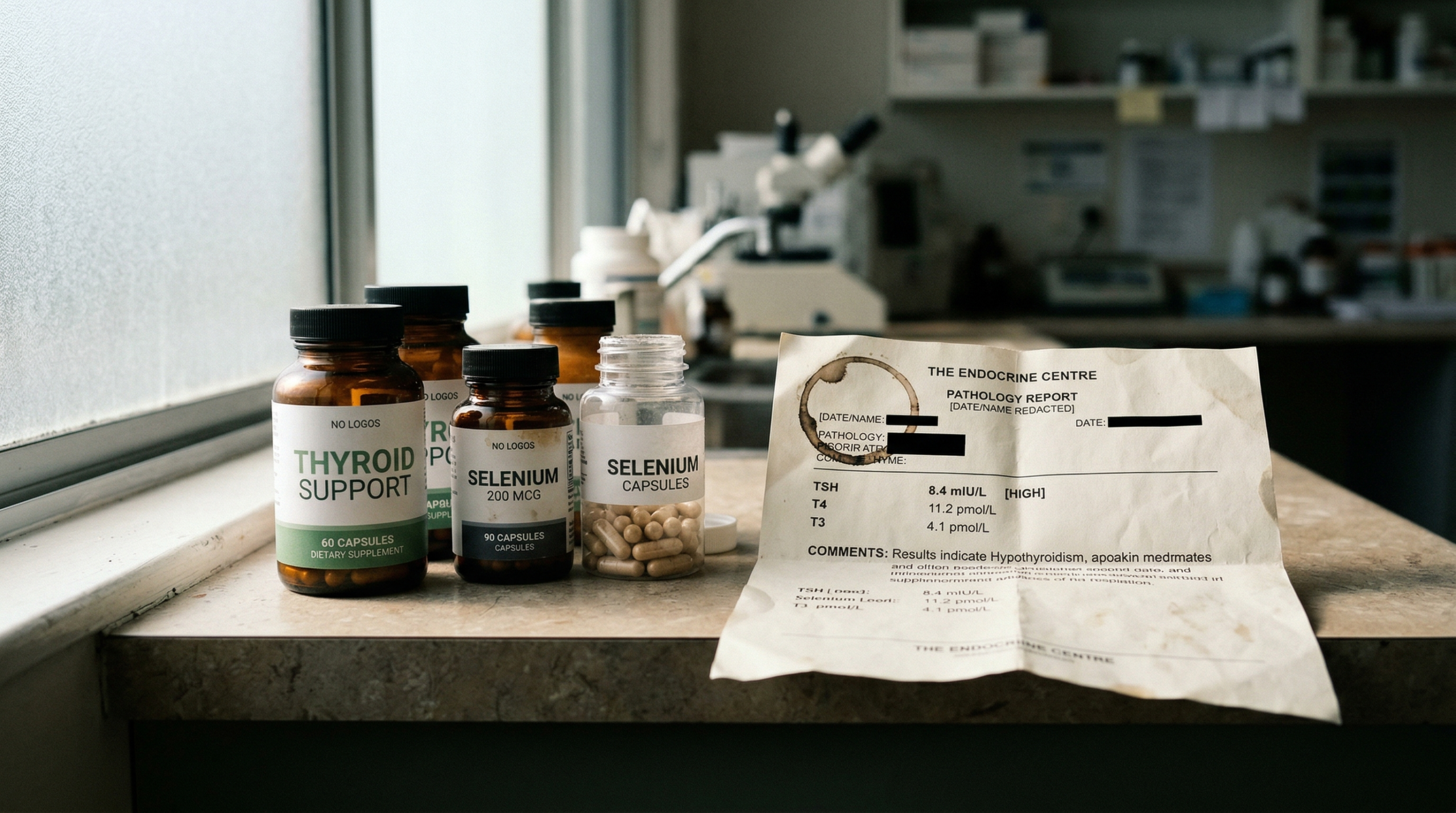
Persistent fatigue, brain fog, weight changes, and low mood may occur even when thyroid-stimulating hormone (TSH) levels appear normal on standard pathology testing. Thyroid function depends on multiple factors beyond TSH, including T4-to-T3 conversion by deiodinase enzymes, selenium and zinc availability, hypothalamic-pituitary-thyroid (HPT) axis regulation, and thyroid-binding globulin (TBG) dynamics. A comprehensive thyroid panel may help identify subclinical dysfunction that routine screening can miss (1–3).
At a Glance
- TSH screening alone may not detect thyroid dysfunction occurring at the level of hormone conversion, transport, or receptor sensitivity (2,4).
- Deiodinase enzymes in the liver and gut convert inactive T4 into active triiodothyronine (T3), and this process can be impaired by nutrient deficiencies or inflammation (8–10).
- Selenium, zinc, and iron are critical cofactors for thyroid peroxidase (TPO) activity and deiodinase function (11–13).
- Chronic stress may increase reverse T3 (rT3) production via non-thyroidal illness syndrome (NTIS), reducing active hormone availability (14–16).
- Elevated oestrogen can raise thyroid-binding globulin (TBG), lowering free T4 and free T3 levels without changing TSH (17,18).
The Core Concept: Why “Normal” Thyroid Tests Can Miss Dysfunction
The thyroid gland produces hormones that regulate basal metabolic rate, thermogenesis, neurological development, and mitochondrial energy production throughout the body. While TSH, secreted by the anterior pituitary gland, is commonly used as a first-line screening tool, it reflects hypothalamic-pituitary-thyroid (HPT) axis signalling rather than thyroid hormone activity at the cellular level (2,4). For an overview of thyroid physiology and common patterns of dysfunction, see our thyroid health resource.
For thyroid hormones to exert their effects, they must be synthesised in the thyroid follicular cells, converted from T4 to T3 by selenoprotein deiodinase enzymes, transported via binding proteins including TBG and transthyretin, and utilised at nuclear thyroid hormone receptors (TR-alpha and TR-beta). Disruption at any of these stages may contribute to symptoms, even when TSH remains within laboratory reference ranges (3,5).
Deeper Drivers of Thyroid Dysfunction
Iodine Status and Thyroid Hormone Production
Iodine is an essential trace element required for the biosynthesis of thyroxine (T4) and triiodothyronine (T3) within the thyroid follicular cells. The World Health Organization (WHO) recognises iodine deficiency as the most common preventable cause of thyroid dysfunction globally. Inadequate iodine intake may impair thyroid peroxidase (TPO)-mediated hormone production, while excessive intake may exacerbate autoimmune thyroiditis in genetically susceptible individuals (6,7).
Impaired Conversion of T4 to Active T3
Approximately 80% of circulating T3 is produced outside the thyroid gland through enzymatic deiodination, primarily by type 1 deiodinase (D1) in the liver and type 2 deiodinase (D2) in the gut, skeletal muscle, and central nervous system. Factors such as selenium and zinc deficiency, systemic inflammation, hepatic impairment, and gastrointestinal dysbiosis may reduce conversion efficiency (8–10). The role of the gut microbiome in thyroid hormone metabolism is explored further in our gut microbiome overview.
Key Nutrient Deficiencies
Several micronutrients serve as essential cofactors in thyroid hormone synthesis, conversion, and receptor binding:
| Nutrient | Role in Thyroid Function | Key Mechanism |
|---|---|---|
| Selenium | Supports deiodinase enzymes (D1, D2, D3) and glutathione peroxidase (GPx) | Protects thyroid tissue from hydrogen peroxide-mediated oxidative damage (11) |
| Zinc | Contributes to thyroid hormone synthesis and nuclear receptor activity | Required for T3 binding to thyroid hormone response elements (TREs) (12) |
| Iron | Required for thyroid peroxidase (TPO) enzymatic activity | Iron deficiency anaemia may impair iodine utilisation and hormone synthesis (13) |
Stress Physiology and Reverse T3
Chronic activation of the hypothalamic-pituitary-adrenal (HPA) axis may alter thyroid hormone metabolism through a process described in the literature as non-thyroidal illness syndrome (NTIS), also known as euthyroid sick syndrome. This involves increased conversion of T4 into reverse T3 (rT3) by type 3 deiodinase (D3). Reverse T3 is a biologically inactive metabolite that can competitively inhibit T3 action at the nuclear receptor level (14–16). Researcher Edith Fliers and colleagues at the University of Amsterdam documented the relationship between illness-related stress and altered deiodinase expression in their 2014 review in Endocrine Reviews (14). This overlap between stress physiology and fatigue patterns is discussed further in our chronic fatigue resource.
Oestrogen and Thyroid Hormone Availability
Elevated circulating oestrogen, whether from oral contraceptive use, hormone replacement therapy, or endogenous overproduction, may increase hepatic synthesis of thyroid-binding globulin (TBG). This reduces the proportion of free, biologically active thyroid hormones (free T4 and free T3) available to tissues (17,18). Bassem Arafah at Case Western Reserve University demonstrated that women with hypothyroidism on oestrogen therapy may require increased thyroxine dosing to maintain adequate free hormone levels (17). Hormone interactions and their systemic effects are also addressed in our mental health and hormones overview.
Assessment: When to Look Beyond TSH
A comprehensive thyroid evaluation is warranted when symptoms of hypothyroidism persist despite TSH falling within the standard laboratory reference range (typically 0.4–4.0 mIU/L). Depending on the clinical picture, assessment may include:
| Test | What It Measures | Clinical Relevance |
|---|---|---|
| Free T3 | Unbound active thyroid hormone | May reveal impaired T4-to-T3 conversion |
| Free T4 | Unbound thyroxine | Indicates thyroid gland output |
| Reverse T3 (rT3) | Inactive T3 metabolite | Elevated levels may suggest stress-mediated conversion |
| TPO & Tg Antibodies | Thyroid autoimmune markers | May indicate Hashimoto’s thyroiditis or Graves’ disease |
| Iron studies, selenium, zinc | Nutrient cofactors | Deficiencies can impair hormone synthesis and conversion |
Management Considerations (Clinician-Guided)
Addressing thyroid dysfunction typically requires an individualised, evidence-informed approach guided by the principles of functional medicine. Nutrient repletion (particularly selenium, zinc, and iron), HPA axis regulation, gut microbiome restoration, hepatic detoxification support, and oestrogen metabolism optimisation are best guided by clinical assessment and laboratory findings rather than applied universally (6,11,14). The American Thyroid Association (ATA) recommends individualised treatment plans based on comprehensive evaluation rather than TSH-only protocols.
Next Steps
- Request a comprehensive thyroid panel: If standard TSH testing has been inconclusive, ask about free T3, free T4, reverse T3, and thyroid antibodies (TPO and thyroglobulin) to get a fuller picture of thyroid function.
- Assess nutrient status: Have iron studies, selenium, and zinc levels checked, as deficiencies in these nutrients can impair thyroid peroxidase activity, deiodinase function, and hormone conversion.
- Book a functional assessment: If thyroid-related symptoms persist despite standard testing, a broader functional medicine evaluation may help identify contributing factors and guide a more targeted management strategy.
Frequently Asked Questions
Key Insights
- Thyroid-related symptoms can occur despite “normal” TSH screening results due to dysfunction at the level of hormone conversion, transport, or receptor sensitivity
- Deiodinase enzymes (D1, D2, D3), nutrient cofactors, HPA axis activity, and thyroid-binding globulin (TBG) levels all influence thyroid hormone bioavailability
- A broader assessment including free T3, reverse T3, TPO antibodies, and thyroglobulin antibodies may help clarify unexplained symptoms
- Selenium, zinc, and iron are critical micronutrients for thyroid peroxidase activity and deiodinase-mediated hormone conversion
- Chronic stress can trigger non-thyroidal illness syndrome (NTIS), increasing reverse T3 production and reducing active thyroid hormone at the tissue level
Citable Takeaways
- Approximately 80% of circulating T3 is produced outside the thyroid gland through peripheral deiodination, primarily by type 1 and type 2 deiodinase enzymes in the liver and gut (Peeters et al., Endocrine Reviews, 2003).
- Selenium is required for the function of all three deiodinase isoforms (D1, D2, D3) and for glutathione peroxidase-mediated protection of thyroid tissue from oxidative damage (Ventura et al., European Thyroid Journal, 2017).
- Iron deficiency may impair thyroid peroxidase (TPO) activity, reducing the efficiency of iodine organification and thyroid hormone synthesis (Beard et al., American Journal of Clinical Nutrition, 1990).
- Chronic physiological stress can increase type 3 deiodinase expression, shifting T4 metabolism toward inactive reverse T3 production, a pattern described as non-thyroidal illness syndrome (Fliers et al., Endocrine Reviews, 2014).
- Oestrogen therapy may increase hepatic thyroid-binding globulin (TBG) synthesis, reducing free T4 and free T3 availability and potentially increasing thyroxine dose requirements in hypothyroid women (Arafah, Annals of Internal Medicine, 2001).
- The World Health Organization recognises iodine deficiency as the most common preventable cause of thyroid dysfunction globally, though excess iodine intake may exacerbate autoimmune thyroiditis (Zimmermann, Endocrine Reviews, 2009).
Get Clarity on Your Thyroid Health
If you are experiencing persistent fatigue, brain fog, or weight changes despite “normal” thyroid tests, a functional medicine assessment can help uncover what standard testing may miss. At Elemental Health and Nutrition, we investigate the deeper drivers of thyroid dysfunction — from nutrient status and deiodinase activity to HPA axis regulation and oestrogen metabolism — to help you feel like yourself again.
References
- Wiersinga WM. Thyroid hormone replacement therapy. Horm Res Paediatr. 2011;76 Suppl 1:74-8. https://doi.org/10.1159/000330225
- Hoermann R, Midgley JEM. TSH measurement and its limitations. Clin Endocrinol (Oxf). 2012 Jul;77(1):1-8. https://doi.org/10.1111/j.1365-2265.2012.04345.x
- Escobar-Morreale HF et al. Thyroid hormone replacement therapy: limitations of TSH. J Clin Endocrinol Metab. 2005 Jul;90(7):4241-3. https://doi.org/10.1210/jc.2005-0455
- Spencer CA et al. Clinical utility of TSH measurement. Thyroid. 2007 Sep;17(9):811-21. https://doi.org/10.1089/thy.2007.0057
- Bianco AC et al. Type 3 deiodinase: role in thyroid hormone action and metabolism. Endocr Rev. 2019 Apr 1;40(2):523-44. https://doi.org/10.1210/er.2018-00191
- Zimmermann MB. Iodine deficiency. Endocr Rev. 2009 Jun;30(4):376-408. https://doi.org/10.1210/er.2009-0011
- Leung AM et al. Iodine intake and autoimmune thyroid disease. Thyroid. 2012 Nov;22(11):1193-8. https://doi.org/10.1089/thy.2012.0244
- Peeters RP et al. Thyroid hormone metabolism. Endocr Rev. 2003 Dec;24(6):823-50. https://doi.org/10.1210/er.2003-0003
- Virili C et al. Gut microbiota and thyroid hormone metabolism. Thyroid. 2018 Dec;28(12):1549-59. https://doi.org/10.1089/thy.2018.0252
- Chiovato L et al. Peripheral thyroid hormone metabolism. Best Pract Res Clin Endocrinol Metab. 2019 Dec;33(6):101335. https://doi.org/10.1016/j.beem.2019.101335
- Ventura M et al. Selenium and thyroid disease: from pathophysiology to treatment. Eur Thyroid J. 2017 Apr;6(2):63-74. https://doi.org/10.1159/000456877
- Bonofiglio D et al. Zinc and thyroid function. Biol Trace Elem Res. 2013 Nov;156(1-3):1-8. https://doi.org/10.1007/s12011-013-9796-5
- Beard JL et al. Iron deficiency alters thyroid hormone metabolism. Am J Clin Nutr. 1990 May;51(5):828-33. https://doi.org/10.1093/ajcn/51.5.828
- Fliers E et al. Stress, illness, and thyroid hormone metabolism. Endocr Rev. 2014 Apr;35(2):271-93. https://doi.org/10.1210/er.2013-1091
- De Groot LJ. Non-thyroidal illness syndrome. In: Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. https://www.ncbi.nlm.nih.gov/books/NBK285560/
- Peeters RP. Reverse T3 and thyroid hormone economy. Best Pract Res Clin Endocrinol Metab. 2017 Dec;31(6):559-70. https://doi.org/10.1016/j.beem.2017.11.004
- Arafah BM. Increased need for thyroxine in women with hypothyroidism during estrogen therapy. Ann Intern Med. 2001 Mar 6;134(5):345-50. https://doi.org/10.7326/0003-4819-134-5-200103060-00006
- Glinoer D. Thyroid regulation during pregnancy. Thyroid. 2001 May;11(5):437-45. https://doi.org/10.1089/105072501300176353






