Hashimoto’s Thyroiditis: A Functional Nutrition Approach

Quick Answer
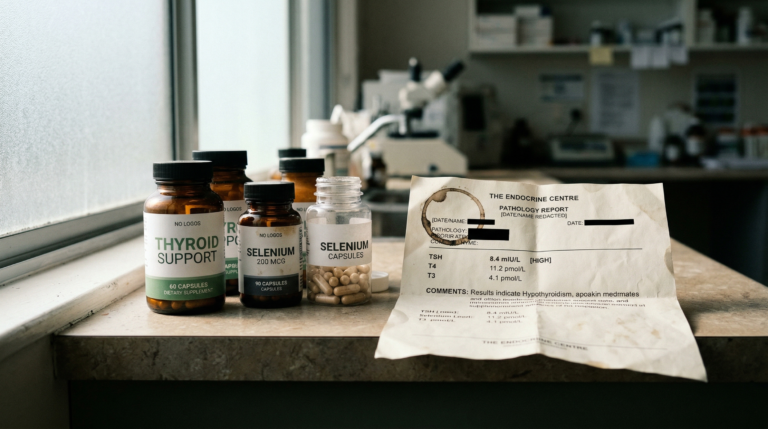
Hashimoto’s thyroiditis is an autoimmune condition in which anti-thyroid peroxidase (anti-TPO) antibodies may gradually damage thyroid tissue, potentially reducing thyroxine (T4) and triiodothyronine (T3) production. A functional medicine approach may complement conventional levothyroxine therapy by addressing modifiable factors such as selenium status, gut microbiome integrity, gluten sensitivity, vitamin D levels, and hypothalamic-pituitary-adrenal (HPA) axis dysregulation that can influence thyroid autoimmunity.
At a Glance
- Hashimoto’s thyroiditis is the most common cause of hypothyroidism, driven by autoimmune destruction of thyroid tissue rather than simple glandular underperformance.
- Anti-TPO antibodies may be elevated years before TSH or thyroid hormone levels become abnormal, making early antibody testing potentially valuable for detection.
- Selenium supplementation has been associated with reductions in anti-TPO antibody levels in some individuals with Hashimoto’s thyroiditis.
- Approximately 70% of the body’s immune tissue resides in the gastrointestinal tract, and gut dysbiosis may contribute to increased intestinal permeability linked to autoimmune activation.
- Vitamin D deficiency is commonly observed in autoimmune thyroid disease and may be associated with increased antibody levels and disease severity.
Hashimoto’s Thyroiditis Is an Autoimmune Attack on Thyroid Tissue
Hashimoto’s thyroiditis, first described by Japanese physician Hakaru Hashimoto in 1912, is an autoimmune condition in which the immune system mistakenly targets thyroid tissue, leading to gradual destruction of the gland and reduced thyroid hormone output over time (1). This autoimmune process may contribute to symptoms including fatigue, cold intolerance, weight gain, low mood, hair thinning, and cognitive changes often described as “brain fog.”
In many cases, early autoimmune activity may not be detected through thyroid-stimulating hormone (TSH) testing alone. Thyroid antibodies, particularly anti-thyroid peroxidase (anti-TPO) antibodies, often rise years before measurable changes in thyroid hormone levels occur (2). A deeper understanding of thyroid autoimmunity is discussed in our overview of thyroid dysfunction and functional testing.
TSH Alone May Miss Active Thyroid Autoimmunity
TSH reflects pituitary signalling rather than thyroid function itself and does not directly assess autoimmune activity. A comprehensive thyroid panel may provide a more complete clinical picture.
| Marker | What It Measures | Clinical Relevance to Hashimoto’s |
|---|---|---|
| TSH | Pituitary signalling to thyroid | May remain normal during early autoimmune activity |
| Free T4 (thyroxine) | Unbound circulating T4 | Indicates thyroid hormone production capacity |
| Free T3 (triiodothyronine) | Active thyroid hormone | May reveal impaired T4-to-T3 conversion |
| Anti-TPO antibodies | Autoimmune activity against thyroid peroxidase enzyme | Primary marker for Hashimoto’s thyroiditis |
| Anti-thyroglobulin antibodies | Autoimmune activity against thyroglobulin protein | Secondary antibody marker for thyroid autoimmunity |
These additional markers, including Free T4, Free T3, anti-TPO antibodies, and anti-thyroglobulin antibodies, may provide a more complete picture of autoimmune thyroid disease and its progression (3).
Gluten Sensitivity, Gut Dysbiosis, and Chronic Stress May Influence Thyroid Autoimmunity
Gluten Sensitivity and Molecular Mimicry
Gluten sensitivity is more prevalent in individuals with autoimmune thyroid disease compared to the general population (4). Research by Alessio Fasano and colleagues has suggested that in susceptible individuals, gluten-derived gliadin peptides may promote immune activation through a process called molecular mimicry, where gliadin’s structural similarity to thyroid tissue may confuse the immune system. This mechanism has been associated with increased thyroid antibody levels and symptom burden (5).
Gut Dysbiosis and Intestinal Permeability
The gastrointestinal tract plays a central role in immune regulation, with approximately 70% of gut-associated lymphoid tissue (GALT) residing within the gut (6). Disruptions in the gut microbiome, referred to as gut dysbiosis, may contribute to increased intestinal permeability, also known as “leaky gut,” where larger molecules pass through the gut lining via compromised tight junctions and interact with the immune system (7).
This mechanism, studied extensively by researchers including Fasano through the zonulin pathway, has been associated with autoimmune conditions, including Hashimoto’s thyroiditis (8). You can explore this relationship further in our detailed guide to the gut microbiome and immune health.
Chronic Stress and the HPA Axis
Chronic stress influences the hypothalamic-pituitary-adrenal (HPA) axis and cortisol regulation, which can interact with thyroid hormone metabolism and immune signalling (9). Persistent stress has been associated with impaired conversion of thyroxine (T4) to the active hormone triiodothyronine (T3) by the deiodinase enzymes, as well as increased pro-inflammatory cytokine activity including interleukin-6 (IL-6) and tumour necrosis factor-alpha (TNF-alpha) (10).
This overlap is frequently observed in people experiencing long-term fatigue and burnout, as discussed in our resource on chronic fatigue and stress-related dysfunction.
Targeted Nutrients May Support Thyroid Function Alongside Medical Care
While thyroid hormone replacement (typically levothyroxine) is often required, a holistic approach aims to support immune balance, nutrient sufficiency, gut integrity, and stress resilience alongside medical management.
Key Nutrient Support for Hashimoto’s Thyroiditis
| Nutrient | Role in Thyroid Health | Research Association |
|---|---|---|
| Selenium | Required for glutathione peroxidase activity and thyroid hormone metabolism via deiodinase enzymes | Supplementation associated with reduced anti-TPO antibody levels in some individuals (11) |
| Zinc | Supports immune regulation, thyroid hormone synthesis, and T4-to-T3 conversion | Suboptimal zinc status observed in individuals with thyroid dysfunction (12) |
| Vitamin D | Modulates immune function via vitamin D receptor (VDR) expression on immune cells | Deficiency commonly observed in autoimmune thyroid disease; associated with increased disease severity (13) |
Supplementation should be guided by pathology testing and practitioner oversight to ensure appropriate dosing and safety.
Gut Healing Protocols
Strategies to support gut health may include dietary modification (such as elimination of identified food sensitivities), targeted use of probiotics including Lactobacillus and Bifidobacterium strains, prebiotics, and functional testing such as comprehensive stool analysis to identify microbial imbalances that may influence immune activity and thyroid autoimmunity (14).
Lifestyle Modifications
| Lifestyle Factor | Mechanism | Potential Benefit |
|---|---|---|
| Stress Management (mindfulness, breathing techniques) | May help regulate cortisol output via HPA axis modulation | Supports immune balance and thyroid hormone conversion |
| Restorative Sleep (7-9 hours) | Supports circadian rhythm, melatonin production, and hormonal regulation | May improve immune function and metabolic health |
| Appropriately Dosed Exercise | Supports insulin sensitivity and stress regulation | May improve overall wellbeing when tailored to individual capacity |
Functional Medicine Addresses the Underlying Drivers of Hashimoto’s
Managing Hashimoto’s thyroiditis often requires a personalised, systems-based approach. Functional and nutritional medicine aim to identify and address underlying contributors such as immune triggers, nutrient insufficiencies, gut dysfunction, and chronic stress patterns.
This approach reflects the broader philosophy of Elemental Health and Nutrition, where investigation and individualisation guide long-term health support.
Next Steps
Hashimoto’s thyroiditis is a complex autoimmune condition, but understanding its contributing factors can empower individuals to take an active role in their care.
- Learn: Explore our thyroid and metabolism resources for deeper insight
- Test: Consider comprehensive thyroid and autoimmune testing beyond TSH
Frequently Asked Questions
Key Insights
- Hashimoto’s thyroiditis is an autoimmune disease, not just low thyroid function
- Normal TSH does not rule out Hashimoto’s; anti-TPO antibodies may be elevated years earlier
- Gut health, intestinal permeability, and diet can influence thyroid autoimmunity
- Chronic stress affects both HPA axis function and thyroid hormone conversion
- Selenium, zinc, and vitamin D may support thyroid health when guided by testing
- Functional and nutritional medicine focus on identifying and addressing underlying drivers
Citable Takeaways
- Anti-TPO antibodies may rise years before measurable changes in TSH or thyroid hormone levels occur, making early antibody testing potentially valuable for Hashimoto’s detection (Ralli et al., Autoimmunity Reviews, 2020).
- Approximately 70% of the body’s immune tissue resides in the gut-associated lymphoid tissue (GALT), linking gastrointestinal health to autoimmune regulation (Belkaid & Hand, Cell, 2014).
- Selenium supplementation has been associated with reductions in anti-TPO antibody levels in some individuals with Hashimoto’s thyroiditis, potentially through its role in glutathione peroxidase and deiodinase enzyme activity (Winther et al., Thyroid, 2014).
- Vitamin D deficiency is commonly observed in autoimmune thyroid disease and has been associated with increased disease severity and thyroid antibody levels (Mackawy et al., Endocrine Practice, 2013).
- Gut dysbiosis and increased intestinal permeability via the zonulin pathway have been associated with autoimmune conditions including Hashimoto’s thyroiditis (Fasano, Physiological Reviews, 2011; De Luca & Shoenfeld, Autoimmunity Reviews, 2019).
Get Personalised Support for Your Thyroid Health
If you suspect Hashimoto’s thyroiditis or are looking for a deeper approach to thyroid management, a comprehensive functional assessment can help identify the factors driving your symptoms. At Elemental Health and Nutrition, we combine thorough testing with personalised nutritional and lifestyle strategies to support your thyroid and immune health.
References
- McLeod DS, Cooper DS. The incidence and prevalence of thyroid autoimmunity. Endocrine. 2012;42(2):252-65.
- Ralli M, et al. Hashimoto’s thyroiditis: An update on pathogenesis. Autoimmun Rev. 2020;19(10):102649.
- Biondi B, Wartofsky L. Treatment with thyroid hormone. Endocr Rev. 2014;35(3):433-512.
- Metso S, et al. Gluten-free diet and autoimmune thyroiditis in patients with celiac disease. Scand J Gastroenterol. 2012;47(1):43-8.
- Ventura A, et al. Duration of exposure to gluten and risk of autoimmune disorders. Gastroenterology. 1999;117(2):297-303.
- Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121-41.
- Fasano A. Zonulin and its regulation of intestinal barrier function. Physiol Rev. 2011;91(1):151-75.
- De Luca F, Shoenfeld Y. The microbiome in autoimmune diseases. Autoimmun Rev. 2019;18(1):3-10.
- Chrousos GP. Stress and disorders of the stress system. Nat Rev Endocrinol. 2009;5(7):374-81.
- Wiersinga WM. Smoking and thyroid. Clin Endocrinol. 2013;79(1):145-51.
- Winther KH, et al. ThyPRO has good responsiveness. Thyroid. 2014;24(7):1078-89.
- Krebs NF. Overview of zinc absorption and excretion. J Nutr. 2000;130(5S Suppl):1374S-7S.
- Mackawy AM, et al. Vitamin D deficiency is associated with thyroid autoimmunity. Endocrine Practice. 2013;19(5):779-85.
- Zhao F, et al. Gut microbiota and thyroid disease. Nutrients. 2022;14(21):4499.