Glutathione & Thyroid Health: The Master Antioxidant
Quick Answer
Glutathione is the body’s primary intracellular antioxidant and may play a critical role in thyroid protection. Because thyroid hormone synthesis by thyroid peroxidase (TPO) requires hydrogen peroxide (H2O2), adequate glutathione is needed to neutralise excess reactive oxygen species and protect thyroid follicular cells from oxidative damage and immune-mediated inflammation. Deficiency in glutathione or its cofactors, including selenium and riboflavin, may contribute to the progression of autoimmune thyroid conditions such as Hashimoto’s thyroiditis (1,2).
At a Glance
- Glutathione is the primary intracellular antioxidant that may protect thyroid follicular cells from hydrogen peroxide-mediated oxidative damage during hormone synthesis.
- Reduced glutathione availability has been associated with increased thyroid peroxidase (TPO) antibody levels and Hashimoto’s thyroiditis progression.
- Selenium-dependent glutathione peroxidase (GPx) is the key enzyme responsible for neutralising H2O2 in thyroid tissue, and selenium supplementation has been associated with lower TPO antibody concentrations (Gartner et al., 2002).
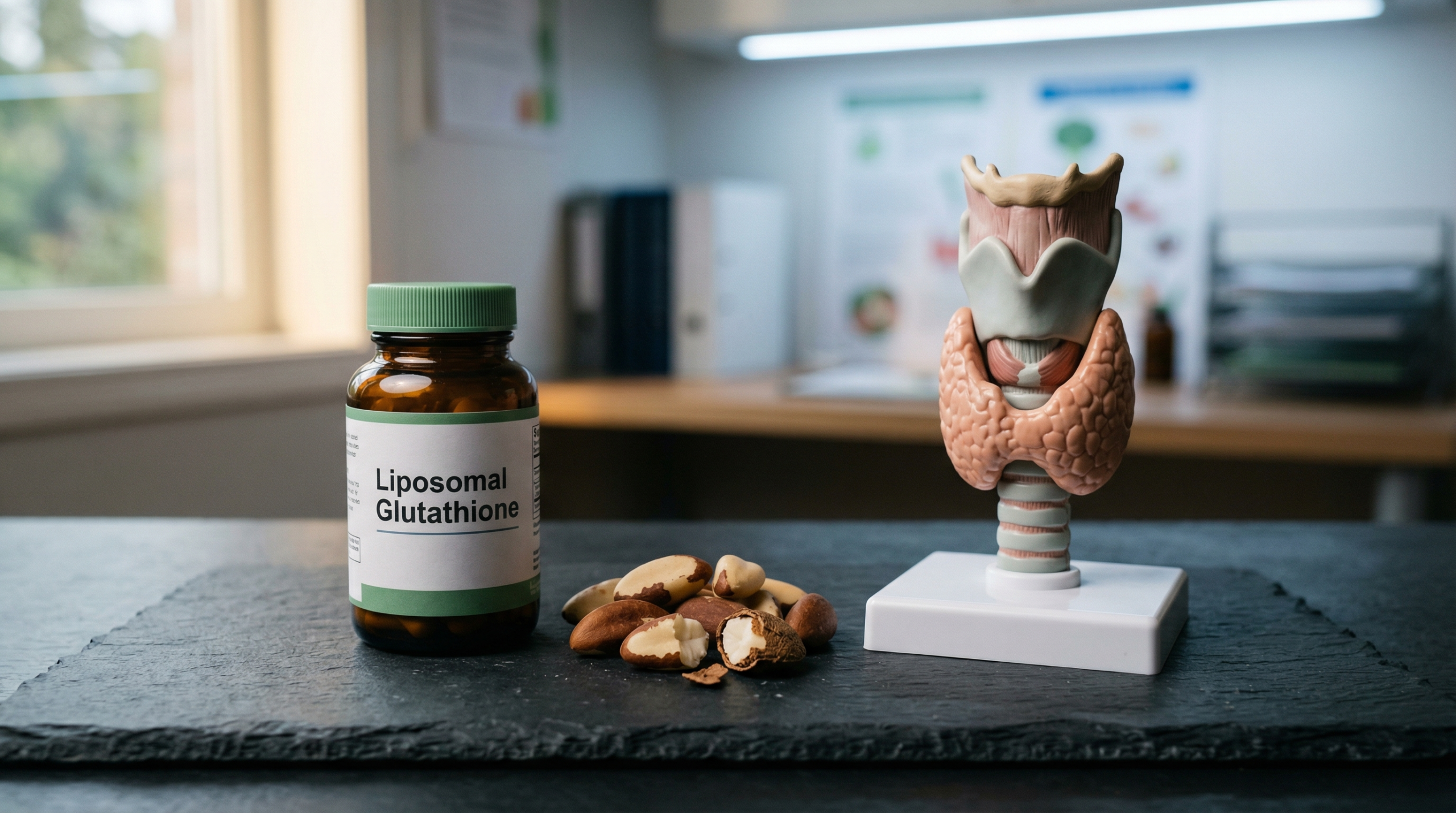
- N-Acetyl Cysteine (NAC) and liposomal glutathione are two commonly used strategies to support glutathione status in clinical practice.
- Riboflavin (vitamin B2) and vitamin D are additional cofactors that may support glutathione recycling and endogenous antioxidant production.
Oxidative Stress and Autoimmune Thyroid Disease
Thyroid follicular cells are among the most metabolically active tissues in the human body, generating significant quantities of hydrogen peroxide (H2O2) during thyroxine (T4) and triiodothyronine (T3) synthesis. When antioxidant capacity, particularly glutathione peroxidase (GPx) activity, is insufficient, oxidative stress can accumulate and damage thyroid proteins, including thyroid peroxidase (TPO). Research published by Song et al. in Frontiers in Endocrinology (2018) suggests this oxidative modification process may contribute to immune recognition and the development of autoimmune thyroid disease, such as Hashimoto’s thyroiditis and Graves’ disease (1–3,13).
Glutathione and Immune Modulation
Depleted intracellular glutathione has been associated with shifts in T-helper cell balance, specifically a Th1/Th2 imbalance, and increased pro-inflammatory cytokine signalling, including elevated tumour necrosis factor-alpha (TNF-alpha) and interleukin-6 (IL-6). Research by Dröge (2002) in Physiological Reviews demonstrated that these immune shifts may increase the likelihood of autoimmune reactivity against thyroid tissue. Supporting glutathione status may therefore assist with immune modulation and reductions in inflammatory activity, rather than directly suppressing immune function (3,4).
Immune balance is also influenced by broader systems involved in immune regulation and inflammation, including gut-associated lymphoid tissue (GALT) and the hypothalamic-pituitary-adrenal (HPA) axis, reinforcing the importance of addressing antioxidant capacity as part of a whole-system approach.
Nutrients Required for Glutathione Function
Glutathione synthesis and recycling depend on several key micronutrients. Deficiency in any of these cofactors may impair the glutathione redox cycle and reduce thyroid antioxidant defence.
| Nutrient | Role in Glutathione Function | Thyroid Relevance |
|---|---|---|
| Selenium | Required cofactor for glutathione peroxidase (GPx), the enzyme that uses glutathione to neutralise H2O2 | Gartner et al. (2002) showed selenium supplementation was associated with reduced TPO antibody levels in autoimmune thyroiditis (4,14,15) |
| Riboflavin (Vitamin B2) | Required for glutathione recycling via glutathione reductase (GR), converting oxidised glutathione (GSSG) back to reduced glutathione (GSH) | Thyroid hormone influences riboflavin activation via flavin adenine dinucleotide (FAD), meaning hypothyroidism may indirectly impair glutathione regeneration (5) |
| Vitamin D | Supports endogenous antioxidant production; deficiency linked to lower glutathione concentrations | Jain et al. (2013) in Molecular and Cellular Biochemistry demonstrated vitamin D deficiency was associated with increased oxidative stress and reduced glutathione levels (6) |
How Glutathione Levels Can Be Supported
Oral glutathione bioavailability is limited due to degradation by gamma-glutamyltransferase (GGT) in the intestinal lumen. Functional strategies therefore focus on improving delivery or supporting endogenous synthesis through the transsulfuration pathway.
| Strategy | Mechanism | Evidence |
|---|---|---|
| Liposomal glutathione | Phospholipid encapsulation improves absorption through the oral and gastrointestinal mucosa | Allen and Bradley (2011) in Journal of Alternative and Complementary Medicine demonstrated improved systemic biomarkers with oral glutathione supplementation (7) |
| N-Acetyl Cysteine (NAC) | Well-studied precursor supplying the rate-limiting amino acid cysteine for glutathione synthesis | Atkuri et al. (2007) described NAC as a safe antidote for cysteine/glutathione deficiency (8) |
| Dietary cysteine sources | Eggs, garlic, onions, cruciferous vegetables, and high-quality whey protein provide substrates for endogenous production | Bounous et al. (1991) and Lands et al. (1999) documented dietary cysteine contributions to glutathione status (9,10) |
| Curcumin | Activates the Nrf2 (nuclear factor erythroid 2-related factor 2) antioxidant response element pathway, upregulating glutathione synthesis enzymes | Balogun et al. (2003) in Biochemical Journal confirmed Nrf2-mediated gene activation by curcumin (11,12) |
Assessing Glutathione Demand
At Elemental Health and Nutrition, assessment extends beyond thyroid-stimulating hormone (TSH) alone and includes functional testing to understand antioxidant demand and methylation capacity:
- Thyroid antibodies (TPO, TgAb): Indicators of immune activity against thyroid tissue and potential markers of oxidative stress burden.
- Gamma-glutamyl transferase (GGT): Low-normal levels may reflect limited glutathione turnover in some individuals; elevated levels may indicate increased oxidative demand.
- Homocysteine and methylation markers: The transsulfuration pathway connects methylation to glutathione synthesis, making these markers relevant to antioxidant assessment.
- Clinical patterns: Findings such as early greying may reflect oxidative stress but are considered observational rather than diagnostic.
Frequently Asked Questions
Key Insights
- Thyroid hormone synthesis generates hydrogen peroxide, increasing the need for antioxidant protection via glutathione peroxidase (1,2).
- Glutathione is central to protecting thyroid follicular cells from oxidative and immune-mediated damage (3).
- Selenium, riboflavin, and vitamin D are required cofactors for glutathione function and recycling (4–6).
- Targeted nutritional and functional strategies, including NAC and liposomal glutathione, may support thyroid resilience in autoimmune conditions.
Citable Takeaways
- Glutathione peroxidase (GPx) is the primary enzyme responsible for neutralising hydrogen peroxide generated during thyroid hormone synthesis, and its activity depends on adequate selenium status (Schomburg, 2011; Rayman, 2012).
- Selenium supplementation was associated with decreased thyroid peroxidase antibody concentrations in patients with autoimmune thyroiditis, according to Gartner et al. (2002) in the Journal of Clinical Endocrinology and Metabolism.
- Depleted intracellular glutathione has been linked to Th1/Th2 immune imbalance and increased pro-inflammatory cytokine activity, which may contribute to autoimmune thyroid disease progression (Dröge, 2002).
- Vitamin D deficiency was associated with increased oxidative stress and reduced glutathione concentrations in a study by Jain et al. (2013) published in Molecular and Cellular Biochemistry.
- Curcumin may upregulate glutathione synthesis enzymes through activation of the Nrf2 antioxidant response element pathway (Balogun et al., 2003).
- N-Acetyl Cysteine (NAC) provides the rate-limiting amino acid cysteine for endogenous glutathione synthesis and has been described as a safe intervention for glutathione deficiency (Atkuri et al., 2007).
Protect Your Thyroid from the Inside Out
If you have thyroid antibodies or persistent symptoms despite “normal” blood tests, a functional assessment may help identify underlying oxidative and immune drivers. At Elemental Health and Nutrition, we take a personalised, evidence-based approach to thyroid health that goes beyond TSH alone.
Next Steps
- Assess thyroid antibodies: Request a comprehensive thyroid panel including TPO and TgAb antibodies to identify immune activity against thyroid tissue.
- Support antioxidant capacity: Optimise selenium, riboflavin, and vitamin D status to support glutathione function and reduce oxidative stress.
- Consider glutathione support: If antioxidant demand is high, liposomal glutathione or NAC supplementation may be appropriate under clinical guidance.
References
- Sugawara M et al. Hydrogen peroxide toxicity in human thyroid cells. Autoimmunity. 1999;30(4):241-50. https://doi.org/10.3109/08916939908999846
- Valdivia A et al. Oxidative stress in thyroid hormone synthesis: a review. Endocr Rev. 2016 Jun;37(3):259-278. https://doi.org/10.1210/er.2015-1138
- Dröge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002 Jan;82(1):47-95. https://doi.org/10.1152/physrev.00018.2001
- Gärtner R et al. Selenium supplementation in patients with autoimmune thyroiditis decreases thyroid peroxidase antibodies concentrations. J Clin Endocrinol Metab. 2002 Apr;87(4):1687-91. https://doi.org/10.1210/jcem.87.4.8421
- Cimino JA et al. Riboflavin and thyroid hormone interactions. J Endocrinol. 1987 Jul;114(1):117-22. https://doi.org/10.1677/joe.0.1140117
- Jain SK et al. Vitamin D deficiency is associated with low glutathione levels and increased oxidative stress in type 2 diabetic patients. Mol Cell Biochem. 2013 Jun;378(1-2):1-8. https://doi.org/10.1007/s11010-013-1593-9
- Allen J, Bradley RD. Effects of oral glutathione supplementation on systemic oxidative stress biomarkers in human volunteers. J Altern Complement Med. 2011 Sep;17(9):827-33. https://doi.org/10.1089/acm.2010.0716
- Atkuri KR et al. N-Acetylcysteine, a safe antidote for cysteine/glutathione deficiency. Curr Opin Pharmacol. 2007 Aug;7(4):355-9. https://doi.org/10.1016/j.coph.2007.04.005
- Bounous G et al. Whey proteins in cancer prevention. Cancer Lett. 1991 May 8;57(2):91-4. https://doi.org/10.1016/0304-3835(91)90072-9
- Lands LC et al. Dietary cysteine supplementation and glutathione status. J Nutr. 1999;129(7):1396-400. https://doi.org/10.1093/jn/129.7.1396
- Balogun E et al. Curcumin activates the haem oxygenase-1 gene via regulation of Nrf2 and the antioxidant-responsive element. Biochem J. 2003 May 1;371(Pt 3):887-95. https://doi.org/10.1042/BJ20021619
- Mahmoudi H et al. Curcumin and antioxidant gene regulation: a review. Phytother Res. 2019;33(5):1195-1206. https://doi.org/10.1002/ptr.6329
- Song Y et al. Oxidative stress and autoimmune thyroid disease. Front Endocrinol (Lausanne). 2018 Oct 22;9:598. https://doi.org/10.3389/fendo.2018.00598
- Rayman MP. Selenium and human health. Lancet. 2012 Mar 31;379(9822):1256-68. https://doi.org/10.1016/S0140-6736(11)61452-9
- Schomburg L. Selenium, selenoproteins and the thyroid gland: interactions in health and disease. Nat Rev Endocrinol. 2011 Oct 11;8(3):160-71. https://doi.org/10.1038/nrendo.2011.174