Iodine, TSH & Thyroid Physiology: What You Need to Know

Quick Answer
Thyroid-stimulating hormone (TSH), produced by the anterior pituitary gland, regulates iodine uptake via the sodium-iodide symporter (NIS) and controls thyroid hormone synthesis. When individuals with iodine deficiency begin iodine supplementation, TSH may temporarily rise as the hypothalamic-pituitary-thyroid (HPT) axis recalibrates to restored iodine availability. This adaptive TSH elevation does not automatically indicate hypothyroidism, particularly when Free T4 and Free T3 remain within reference ranges.
Research by Zimmermann (2009) and Leung and Braverman (2014) shows iodine repletion can alter TSH dynamics during this adaptive phase, especially in previously deficient individuals. Symptoms such as fatigue may improve even while TSH remains transiently elevated (1-4).
At a Glance
- TSH is a pituitary signalling hormone, not a thyroid hormone itself; it regulates iodine uptake and thyroid hormone production via the sodium-iodide symporter (NIS).
- Iodine repletion in deficient individuals may trigger a transient TSH elevation lasting weeks to months as the HPT axis adapts (Zimmermann, 2009).
- Free T4 and Free T3 levels, alongside clinical symptoms, provide more meaningful assessment than isolated TSH during iodine repletion.
- Cofactors including selenium, zinc, and iron are essential for thyroid peroxidase (TPO) activity and iodothyronine deiodinase-mediated T4-to-T3 conversion.
- Iodine supplementation is not appropriate for individuals with Hashimoto’s thyroiditis, Graves’ disease, or nodular thyroid conditions without clinical supervision.
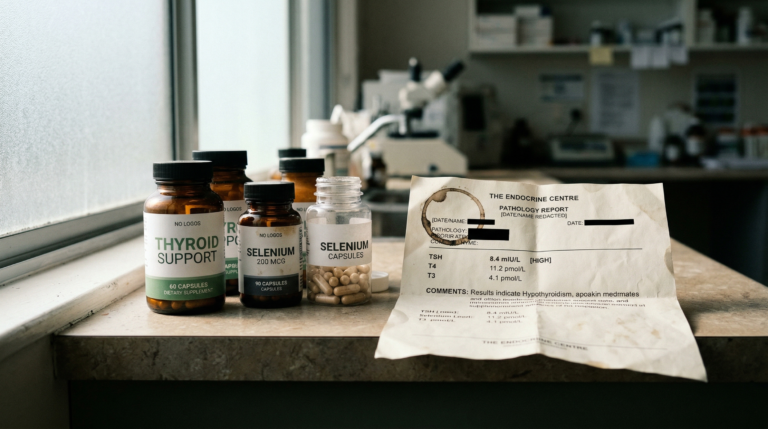
If you’ve been dealing with fatigue, unexplained weight changes, or thinning hair, the thyroid is often part of the conversation. In our Adelaide clinic, one frequently overlooked factor is iodine status. However, when iodine supplementation begins, patients are often surprised to see a sharp rise in their TSH on follow-up testing.
At Elemental Health and Nutrition, we help patients interpret thyroid markers in context — so lab results support clinical decision-making rather than creating unnecessary alarm.
Understanding the Iodine-TSH Relationship
Iodine is an essential trace element required for the biosynthesis of thyroid hormones thyroxine (T4) and triiodothyronine (T3), as confirmed by the World Health Organization (WHO) and the Australian National Health and Medical Research Council (NHMRC).
| Thyroid Hormone | Iodine Atoms | Primary Role |
|---|---|---|
| Thyroxine (T4) | 4 | Prohormone; converted to active T3 by deiodinase enzymes (DIO1, DIO2) |
| Triiodothyronine (T3) | 3 | Biologically active hormone; regulates basal metabolic rate, thermogenesis, and gene expression |
While iodine availability directly affects hormone production, TSH is centrally regulated by the hypothalamus via thyrotropin-releasing hormone (TRH) and may rise during periods of thyroid remodelling or altered iodine handling (1,2).
One key mechanism involves upregulation of the sodium-iodide symporter (NIS), the transport protein responsible for moving iodine into thyroid follicular cells. Research by Eng et al. (1999) demonstrated that increased TSH signalling can enhance NIS expression during iodine repletion, particularly following deficiency. The Wolff-Chaikoff effect, first described by Jan Wolff and Israel Chaikoff in 1948, describes the transient inhibition of thyroid hormone synthesis in response to acute iodine loading (3,4).
Clinical Observation (Non-Generalisable Data)
In our Adelaide clinical setting at Elemental Health and Nutrition, some individuals with low or low-normal Free T4 display biochemical patterns consistent with iodine insufficiency, even when baseline TSH falls within standard Australian population reference ranges (typically 0.4-4.0 mIU/L).
These observations are clinic-specific and not intended as population-level data. They are used to inform individualised assessment, not to infer prevalence or causation. Population studies by Pearce et al. (2016), Andersson et al. (2007), and Bath and Rayman (2013) confirm that iodine deficiency can exist despite apparently normal TSH values (5-7).
The Role of Cofactors in Iodine Utilisation
Adequate cofactor status is critical for safe and effective thyroid hormone metabolism, as demonstrated by Zimmermann and Kohrle (2002) and Arthur and Beckett (1999).
| Cofactor | Mechanism | Key Evidence |
|---|---|---|
| Selenium | Required for iodothyronine deiodinases (DIO1, DIO2, DIO3) that convert T4 to active T3; also supports glutathione peroxidase (GPx), protecting thyroid tissue from oxidative damage | Arthur and Beckett, 1999; Kohrle, 2005 (8,9) |
| Zinc | Involved in thyroid hormone receptor binding, TSH regulation, and TRH synthesis in the hypothalamus | Baltaci et al., 2017 (10) |
| Iron | Essential for thyroid peroxidase (TPO) activity, the enzyme catalysing iodine organification | Beard et al., 1990; Zimmermann and Kohrle, 2002 (11,14) |
| Cortisol (stress marker) | Elevated cortisol may be associated with reduced peripheral T4-to-T3 conversion via suppression of DIO2 activity | Fekete and Lechan, 2014; McEwen, 2007 (12,13) |
Symptoms Commonly Associated With Iodine Deficiency
Iodine deficiency has been associated with impaired thyroid hormone production and metabolic slowing, as documented in the Global Iodine Nutrition Scorecard and research by Laurberg et al. (2010).
- Fatigue and cold intolerance
- Dry skin and hair thinning
- Cognitive slowing or “brain fog”
- Menstrual irregularities and reduced libido
Monitoring During Iodine Repletion
Following iodine introduction, TSH may remain elevated for several months in some individuals while hypothalamic-pituitary-thyroid axis signalling adapts. During this period, Free T4, Free T3, and symptom trends provide more meaningful clinical context than TSH alone, as supported by Kohrle (2005), Leung and Braverman (2014), and Arthur and Beckett (1999) (2,4,9).
Iodine supplementation is not appropriate for all individuals, particularly those with autoimmune thyroid disease (Hashimoto’s thyroiditis, Graves’ disease), nodular thyroid conditions, or iodine sufficiency. Clinical supervision including urinary iodine concentration (UIC) testing and thyroid antibody panels (anti-TPO, anti-thyroglobulin) is essential before and during supplementation.
Next Steps
- Interpret TSH in context: If your TSH has risen after starting iodine, check Free T4 and Free T3 alongside symptom trends rather than reacting to TSH alone.
- Assess cofactor status: Ensure selenium, zinc, and iron levels are adequate to support safe and effective iodine utilisation and deiodinase-mediated T4-to-T3 conversion.
- Seek clinical supervision: Iodine supplementation requires monitoring — particularly if you have autoimmune thyroid disease or nodular thyroid conditions. A functional assessment can help distinguish adaptive changes from clinically significant dysfunction.
Frequently Asked Questions
Key Insights
- TSH is a regulatory signal produced by the anterior pituitary, not a thyroid hormone (1)
- Iodine repletion may be associated with transient TSH elevation in iodine-deficient individuals via NIS upregulation (2-4)
- Free T3 and Free T4 provide essential clinical context during thyroid adaptation (8,9)
- Cofactor sufficiency — selenium, zinc, and iron — is critical for safe and effective iodine utilisation (8-11)
Citable Takeaways
- Iodine deficiency remains a global concern; Zimmermann (2009) reported that nearly 2 billion individuals worldwide have insufficient iodine intake, affecting thyroid hormone synthesis (Endocrine Reviews, 30(4):376-408).
- The sodium-iodide symporter (NIS) is upregulated by TSH signalling during iodine repletion, as demonstrated by Eng et al. (1999) in FRTL-5 thyroid cell models (Endocrinology, 140(8):3404-10).
- Selenium-dependent iodothyronine deiodinases (DIO1, DIO2, DIO3) are essential for T4-to-T3 conversion; Kohrle (2005) confirmed that selenium deficiency may impair peripheral thyroid hormone activation (Thyroid, 15(8):841-53).
- Iron deficiency can reduce thyroid peroxidase (TPO) activity and impair iodine organification, as reported by Beard et al. (1990) in the American Journal of Clinical Nutrition (52(5):813-9).
- Population-level iodine deficiency can persist despite apparently normal TSH values, as documented by Pearce et al. (2016) in the Lancet Diabetes and Endocrinology (4(6):538-50).
- The Wolff-Chaikoff effect, first described in 1948, demonstrates that acute iodine excess transiently inhibits thyroid hormone synthesis — a protective autoregulatory mechanism (Journal of Biological Chemistry, 174(2):555-64).
Confused by Your Thyroid Results?
Thyroid markers are best interpreted in context, not isolation. If your TSH has changed after starting iodine, a functional assessment of iodine status, thyroid hormones, and key cofactors can help distinguish adaptive changes from clinically significant dysfunction. At Elemental Health and Nutrition, we provide personalised, evidence-informed thyroid support in Adelaide.
References
- Zimmermann MB. Iodine deficiency. Endocr Rev. 2009 Jun;30(4):376-408. https://doi.org/10.1210/er.2009-0011
- Leung AM, Braverman LE. Consequences of excess iodine. Nat Rev Endocrinol. 2014 Mar;10(3):136-42. https://doi.org/10.1038/nrendo.2013.251
- Wolff J, Chaikoff IL. Plasma inorganic iodide as a homeostatic regulator of thyroid function. J Biol Chem. 1948 Sep;174(2):555-64. https://doi.org/10.1016/S0021-9258(18)57234-9
- Eng PHK et al. Regulation of the sodium iodide symporter by iodide in FRTL-5 cells. Endocrinology. 1999 Aug;140(8):3404-10. https://doi.org/10.1210/endo.140.8.6897
- Pearce EN et al. Consequences of iodine deficiency and excess in pregnant women: an overview of current knowledge. Lancet Diabetes Endocrinol. 2016 Jun;4(6):538-50. https://doi.org/10.1016/S2213-8587(16)00054-7
- Andersson M et al. Iodine deficiency in Europe: a continuing public health problem. Lancet. 2007 Dec 15;370(9604):1856-60. https://doi.org/10.1016/S0140-6736(07)61786-0
- Bath SC, Rayman MP. Iodine deficiency in the UK: an overlooked issue? Lancet. 2013 Aug 10;382(9891):1154-5. https://doi.org/10.1016/S0140-6736(13)61836-1
- Arthur JR, Beckett GJ. Thyroid function. Br Med Bull. 1999;55(3):658-68. https://doi.org/10.1258/1358869991923948
- Kohrle J. Selenium and the control of thyroid hormone metabolism. Thyroid. 2005 Aug;15(8):841-53. https://doi.org/10.1089/thy.2005.15.841
- Baltaci AK et al. Zinc and thyroid function. Biol Trace Elem Res. 2017 Nov;179(1):1-8. https://doi.org/10.1007/s12011-017-0957-5
- Beard JL et al. Iron deficiency alters thyroid hormone metabolism. Am J Clin Nutr. 1990 Nov;52(5):813-9. https://doi.org/10.1093/ajcn/52.5.813
- Fekete C, Lechan RM. Central regulation of hypothalamic-pituitary-thyroid axis under physiological and pathophysiological conditions. Endocr Rev. 2014 Apr;35(2):159-94. https://doi.org/10.1210/er.2013-1087
- McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007 Jul;87(3):873-904. https://doi.org/10.1152/physrev.00041.2006
- Zimmermann MB, Kohrle J. The impact of iron and selenium deficiencies on iodine and thyroid metabolism. Thyroid. 2002 May;12(5):427-36. https://doi.org/10.1089/105072502760143926
- Laurberg P et al. Iodine intake as a determinant of thyroid disorders in populations. Best Pract Res Clin Endocrinol Metab. 2010 Feb;24(1):13-27. https://doi.org/10.1016/j.beem.2009.08.013